Image credit: Ben McDade / Wellcome Sanger Institute
Discover how the Wellcome Sanger Institute has been at the forefront of spatial research, exploring how trillions of cells organise, communicate and work together to form a human being. For more than 15 years, Sanger scientists have pushed the boundaries of modern genomics, helping to map the human body in greater detail – cell by cell.
Genetics has massively evolved from Gregor Mendel’s 19th-century discovery of inheritance. The structure of DNA was revealed in 1953, enabling molecular biology and genetic engineering. Then, the Human Genome Project in the late 20th century ushered in large-scale genomics, while next-generation sequencing made genome and transcriptome analyses routine. In the 2010s, single-cell sequencing exposed cellular diversity, and spatial transcriptomics now maps gene activity within tissues, bringing unprecedented insight into biology.
Spatial transcriptomics is becoming a vital part of our work here at the Wellcome Sanger Institute, so here we look back at the early adoption of these technologies at Sanger and the components that enabled them to become a part of our core operations.
Advancements in spatial technologies
Our spatial journey started back in the 2010s with the beginning of our efforts on single-cell biology, which involves the study of genes, molecules and activities in each individual cell. This is in contrast to so-called ‘bulk’ approaches where many cells are analysed together. Single-cell analysis helps scientists see differences between cells, understand how they work and find rare cell types. This work was sparked at the time by former Head of the Cellular Genomics programme and Co-Founder of the Human Cell Atlas, Professor Sarah Teichmann, former Associate Faculty, Dr John Marioni, former Group Leader, Dr Thierry Voet and former Head of Research and Development, Dr Harold Swerdlow – leading to the formation of the Sanger-EBI Single-Cell Initiative. This Initiative was set up to develop and apply methods to gain information on the complete genetic contents of single cells in a high-throughput manner.
This work was driven by the availability of single-cell transcriptomic technologies, most importantly the 10x Genomics Chromium platform. This is a droplet-based technology that captures each cell in a tiny droplet with special barcodes to help track which genes are expressed by which cells. This can make it easier to understand how different cells in a tissue work and interact.
Whilst single-cell assays can capture very detailed information about each cell, it was apparent at the time that cells were being taken from their native environment and studied without spatial tissue context. In solid organs where cells are located, the identity of their neighbours, their tissue niche, their microenvironment, their communication – all of these are key determinants of a cell's identity and behaviour. The team and wider single-cell community recognised that spatial resolution was needed to deepen our insights.
At this time, spatial technologies were at very early stages, so the Institute decided to bring in leading researchers to help drive this process. In 2018, Dr Omer Bayraktar joined the Cellular Genomics programme as a Group Leader. Omer had the desire to do spatial transcriptomics to understand cells in their native tissues in the human body with a focus on the brain. During this time, the Human Cell Atlas (HCA) was also kicking off – a project which aims to map all the cells of the human body to understand more about health and disease. This project – co-founded by Sarah Teichmann and strongly driven by research at the Institute – was generating a lot of single-cell data, but the community wanted to start doing spatial too. In response, the High-Throughput Spatial Genomics group was formed at the Institute in 2019 to develop and scale spatial transcriptomics. Their work enables the integration of spatial data with single-cell atlases, helping build detailed, 3D maps of human tissues relevant to health and disease.
In the same year, Dr Kenny Roberts, now Senior Staff Scientist, joined Omer’s team as a postdoc and worked on a project to help set up spatial. They started by doing RNAscope – a commercially available platform that uses single-molecular fluorescence in situ hybridisation (smFISH) to visualise and quantify individual RNA transcripts within cells and tissues using fluorescent probes. They were using this to assay a small number of genes – approximately four to 12 – and generated data that contributed to various early HCA papers.
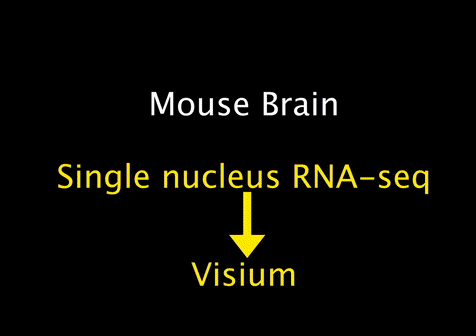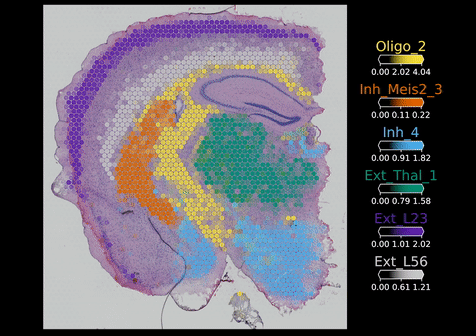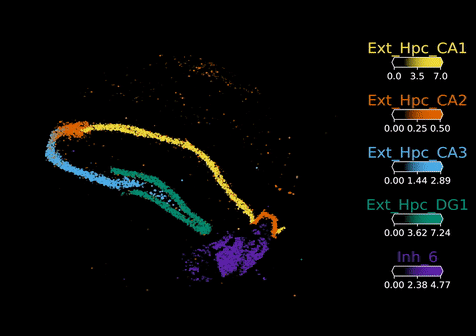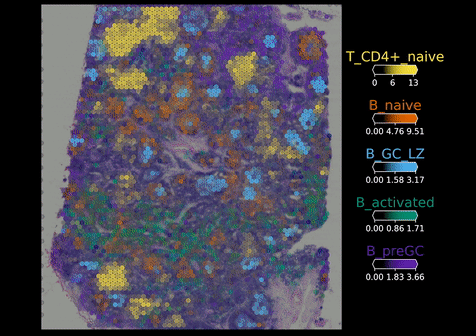
In late 2019, the Institute was among the first in the world to pilot the 10x Genomics Visium assay – the first commercial sequencing-based spatial transcriptomics workflow which allows you to look at the entire transcriptome, so approximately 20,000 genes. This complemented the imaging-based RNAscope approach and by 2020, both platforms were well-established and in widespread use across the Cellular Genomics programme. At the same time, Kenny and colleagues in Omer’s group started setting up in situ sequencing (ISS) – another imaging-based technique developed by Associate Faculty, Professor Mats Nilsson, which visualises larger numbers of RNA molecules in intact tissues or cells. This allowed the team to look at approximately 100 to 200 genes at a time, though it was a very manual process requiring a lot of laboratory work, imaging and image analysis.
In 2022, the Institute began to use CytAssist, which is an instrument developed by 10x Genomics to enhance the capability of the Visium spatial transcriptomic workflows. It facilitates the transfer of probes from standard glass slides to Visium slides and improves the localisation of transcripts.
The following year, a different imaging-based 10x Genomics spatial platform – Xenium – was integrated into Sanger. By 2024, Xenium released an assay – Xenium Prime 5K – that offered the ability to analyse 5,000 genes on samples compared to 500 in the original version.
“When I first joined the Institute five years ago, doing RNAscope was the coolest thing I had ever seen. When Xenium came along and we did the first Xenium run in Europe on a mouse brain sample, I jumped up and down when we saw the data. It was such a paradigm shift in how we work. It was incredible. And now, we are doing 5,000 genes. I never thought it would be possible, so to think about what we could be doing in another five years is exciting.”
Dr Kenny Roberts,
Senior Staff Scientist, Cellular and Gene Editing Research, Wellcome Sanger Institute
“Spatial transcriptomics is integral to the work we do in the lab, and increasingly important across Sanger. Personally, it's been remarkable to witness the pace of technological change in the field. I'm amazed at the ability of my colleagues at the Institute to adapt to this change to increase the resolution and throughput of our tissue analyses. Spatial transcriptomics is inherently collaborative, with expertise covering histology, sequencing, data analysis and more – a fantastic example of the power of team science.”
Dr Omer Bayraktar,
Group Leader, Cellular Genomics programme, Wellcome Sanger Institute
Development of computational tools and infrastructure
Spatial genomics produces large amounts of data – which need to be analysed, visualised and appropriately stored to maximise insights.
To integrate spatial fully, the Informatics and Digital Solutions (IDS team) at Sanger had to establish new forms of data storage that were much better for handling image and spatial transcriptomic data. This saw the emergence of Isilon storage, which stores large amounts of data in a way that is easy to manage and scale.
Once these data are stored, they need to be analysed. But first, the raw data need to be processed to map points like the biology of individual cell types, their interactions and their niches. To support with spatial analysis, researchers in the Cellular Genomics programme started to build a series of computational tools. One of these is Cell2location – developed by Dr Vitali Kleschevnikov, a former graduate student in Omer’s lab – which enables researchers to map individual cell types within tissues by integrating single cell RNA sequencing data with spatial transcriptomics data. This is to overcome the limitations of some existing spatial platforms that do not provide true single-cell resolution.
One of the attractive elements of this data modality to many people, even beyond the genomics community, is the pretty visualisation of cells in tissues. To do this, software is needed to allow scientists to navigate tissue sections, zoom in and out of different parts and search for cell types or genes in an automated way. As such, the Cellular Genomics programme introduced Omero Web Server – a platform used for managing, storing and sharing biological images. This platform, now maintained by the IDS team, allows the Institute to keep all the imaging-based data in one place, and navigate it like you would on Google Maps. Remote access can also be provided to collaborators who can annotate regions to help inform downstream analyses. This platform is mainly for histological stains.
Last year, researchers from Omer and Professor Muzz Haniffa’s (Head of Cellular Genomics, Senior Group Leader and Deputy Director at the Sanger Institute) groups, led by Dr Tong Li, David Horsfall and Daniela Basurto Lozada, also built and published WebAtlas. This is a platform developed to facilitate the sharing and interactive exploration of integrated single-cell and spatial transcriptomic datasets. Unlike Omero, it can look at both image and transcriptomic data to identify boundaries of individual cells and where RNA molecules are located. You can filter and visualise the data based on different cell types or genes.
Making spatial scalable by integrating it into Scientific Operations
The Spatial team, part of Sanger’s Scientific Operations, is a team full of expertise in laboratory techniques associated with spatial genomics and allows researchers to up-scale their projects. RNAscope was the first spatial technique introduced into the Spatial team as a service in the early 2020s and enabled research teams to generate more data at higher throughput. When the Visium and Xenium machines made their way to Sanger, RNAscope work was phased out, and the use of these more advanced technologies increased.
A lot of this was driven by the HCA team who wanted to map the architecture of different human organs and apply this to hundreds and thousands of patient tissue samples. In order to do this, it was vital to integrate these technologies into the Spatial team's offerings. Working with the Head of Cellular and Gene Editing Research, Dr Andrew Bassett’s team and Senior Scientific Manager, Minal Patel’s team, and researchers in the Cellular Genomics programme, Visium and Xenium became available for all of the scientific programmes at Sanger to use.
Now at the Institute, we have five Xenium machines and three CytAssist Visium machines that are run at scale by the Spatial team.
“There has been a lot of great progress made over the last decade in terms of the co-ordination on engagement, demand planning, capacity building, throughput increase, alignment and scaling of the spatial platforms into production. We remain the first in the world table for the amount of 10x Genomics Xenium 5K runs. It is a great example of the impact of close engagement and collaborative working across Sanger’s scientific programmes, Management Operations and Scientific Operations.”
Minal Patel,
Spatial Operations Manager, Spatial team, Wellcome Sanger Institute